
Pega for Life Sciences
Accelerate the development and delivery of life sciences solutions.

"The biggest reason we chose Pega was the security aspect. We could not have companies seeing other companies’ transactions. Pega worked really well for that."
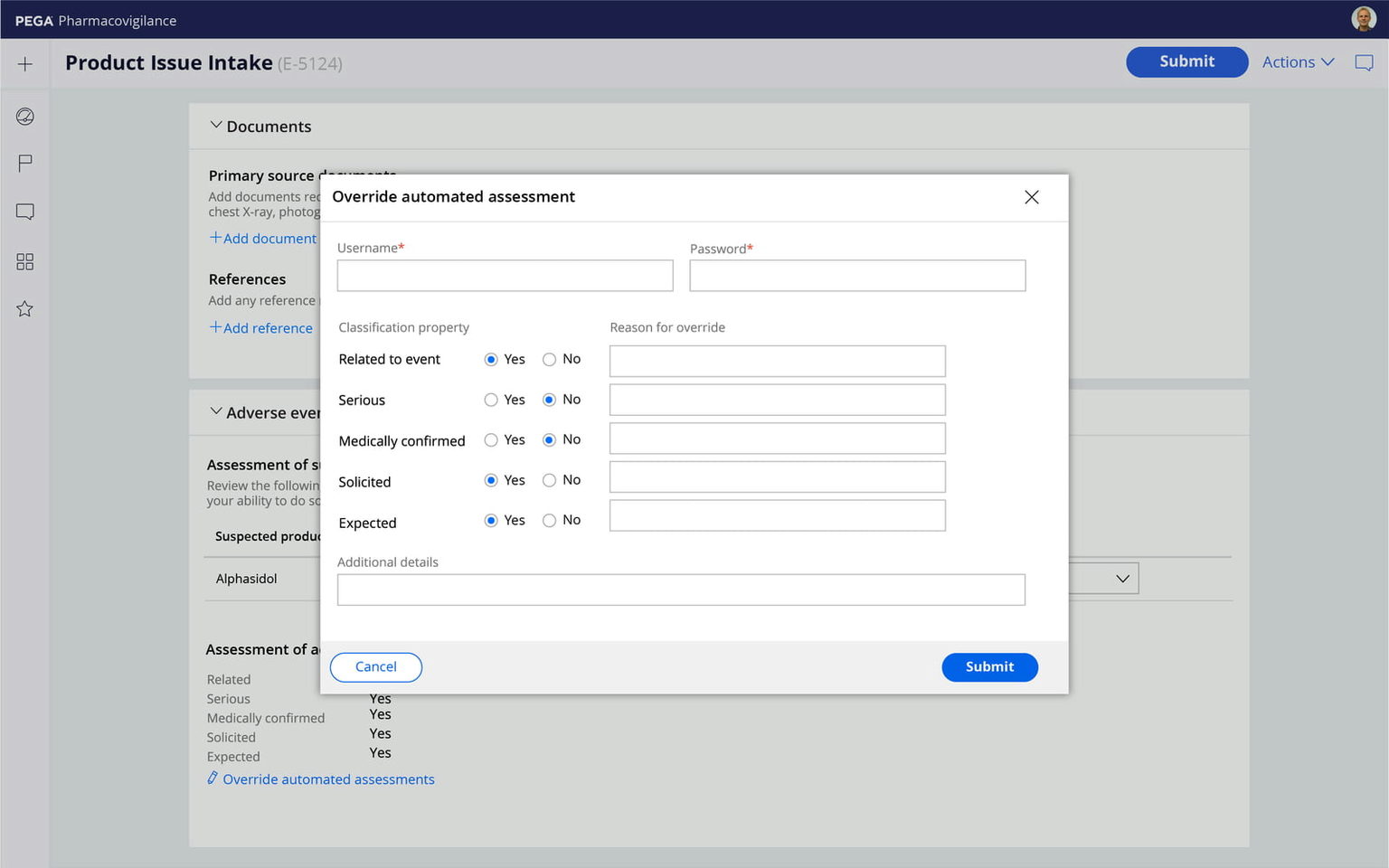
Rapidly add electronic signatures and comprehensive audit trails to any Pega application. An easy-to-use wizard identifies where your applications need to support Part 11 requirements, then automatically configures required modification detection logic, user interface, and audit trail elements.
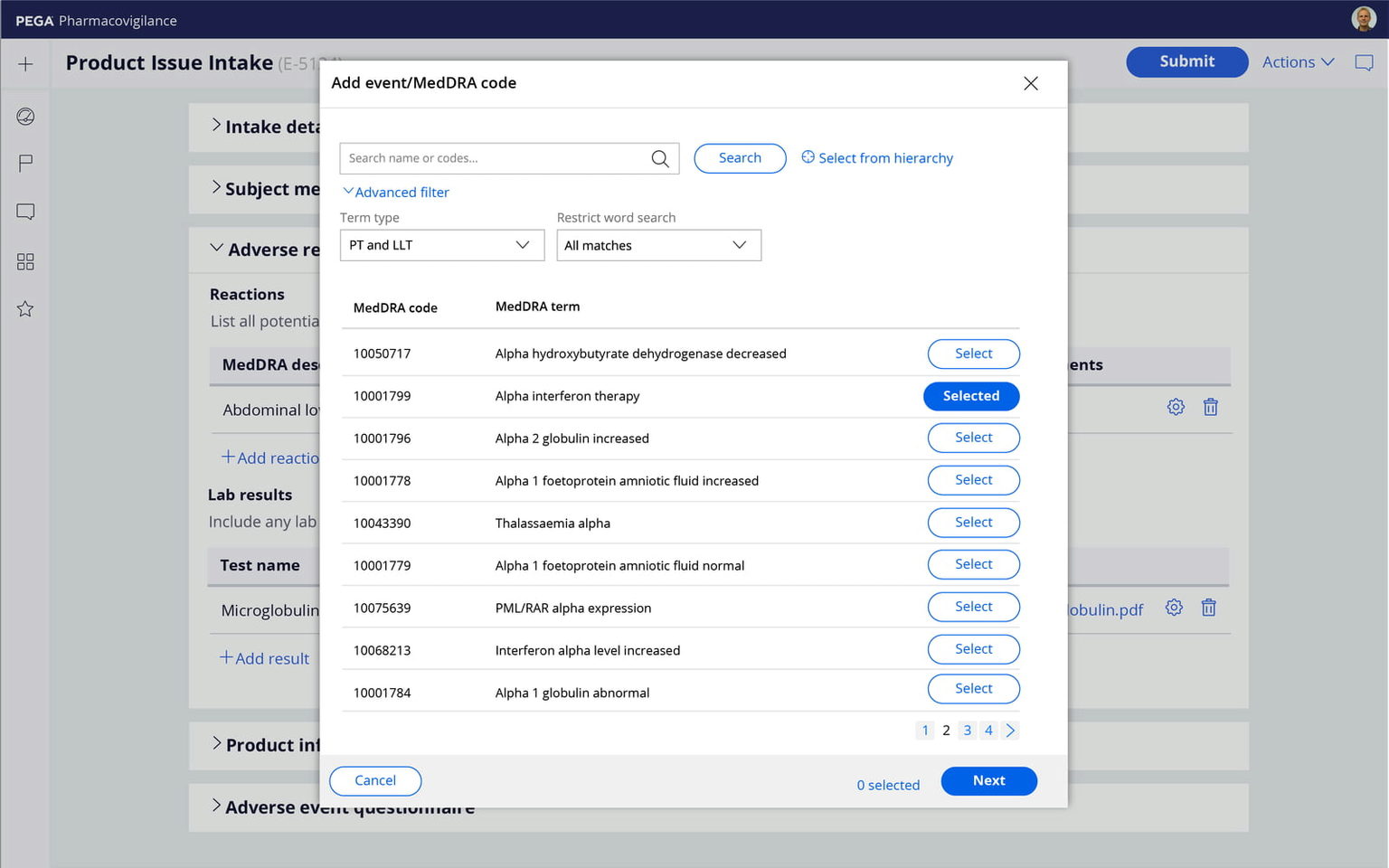
A configurable code list browser supports tree-view navigation and advanced search for hierarchical code lists. The product search component provides easy navigation of internal/external product tables. Out-of-the-box support for MedDRA codes and WHODrug data is also included.
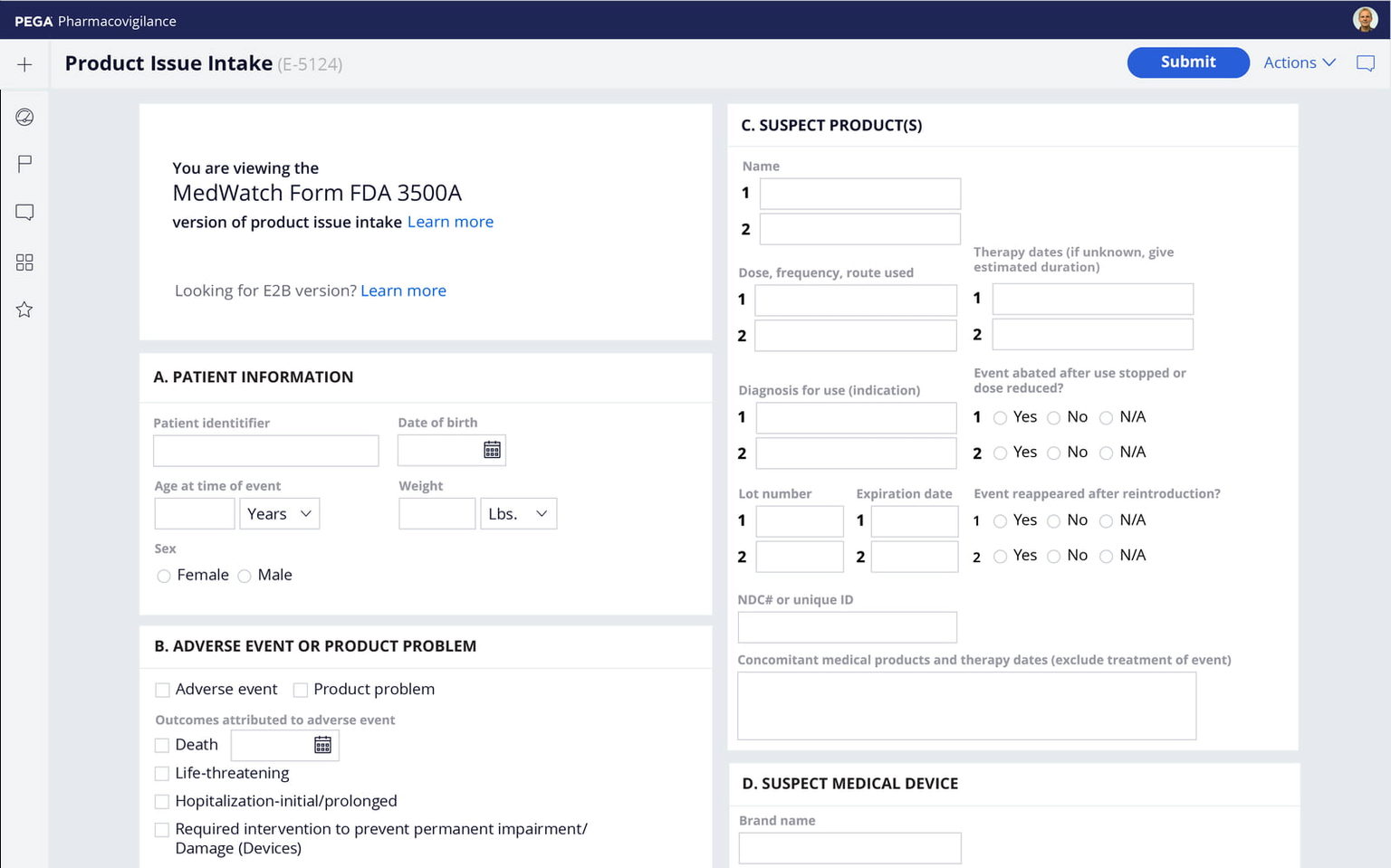
Easily configure PDF generation for regulatory forms and documents. Wizard-based tools let you quickly map data from Pega applications to PDF templates. Preserve the precise look and feel of documents for regulatory content.
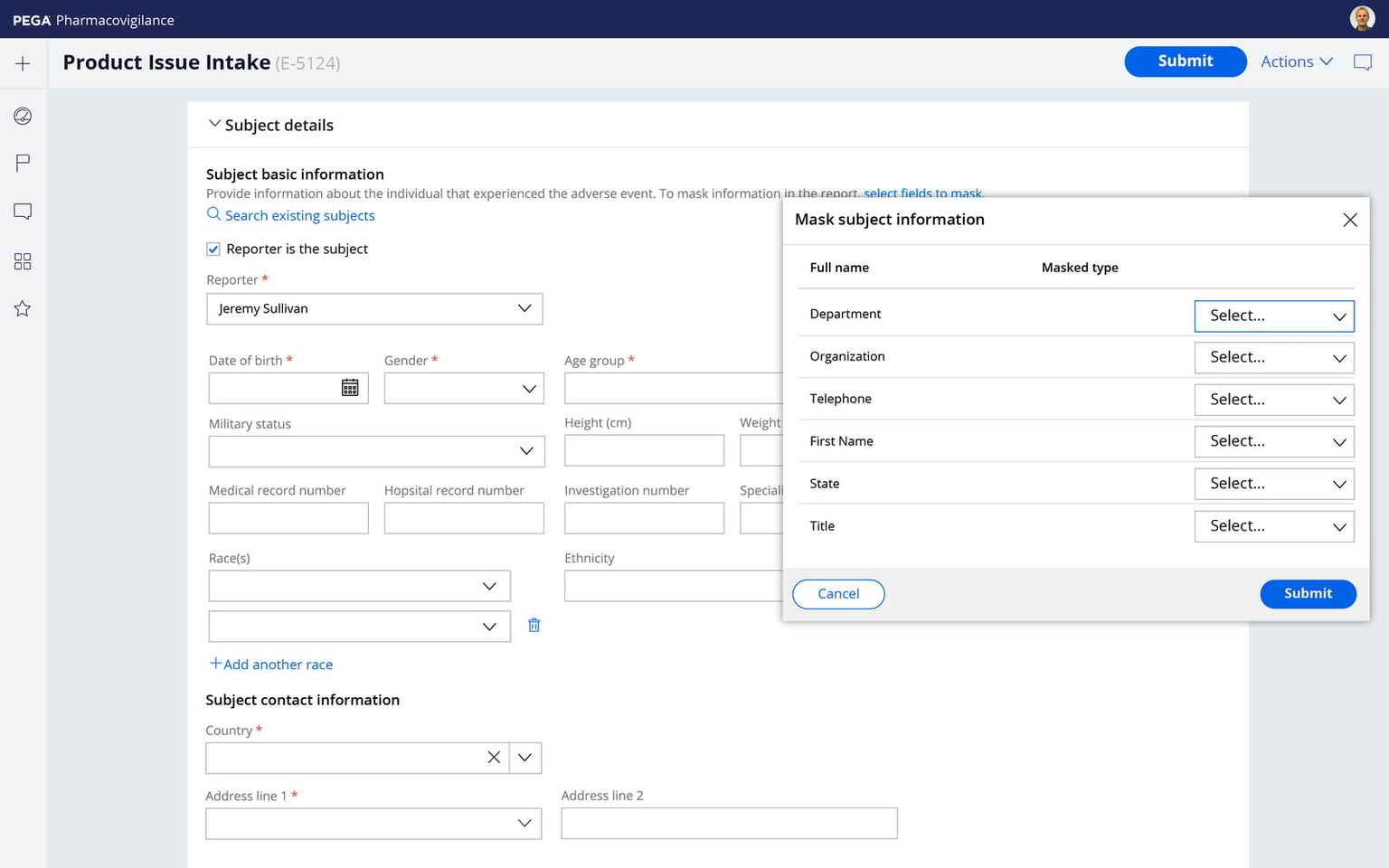
Pega lets you interact securely with external process participants like physicians, investigators, and patients, track interactions progress and completion automatically for multiple participants, and authenticate external users with multi-factor authentication and one-time passwords.
Pega BPM and Case Management
Quickly configure business applications with built-in business process and case management.
Data Access Made Easy
Unique data management capabilities enable you to create a virtualized application data layer.
Pega Cloud
Pega’s scaled, tiered cloud environment supports the Pega Customer Service application.
Customer Case Studies
See how customers are using Pega for Life Sciences to transform their enterprises.
See how Pega can help your business
Talk to an expert and see how Pega can transform your customer experiences with next-generation software applications and solutions.

